Evaluation of Potential Mechanisms of SUDEP in a Mouse Model of Dravet Syndrome
Rationale:
Dravet syndrome (DS) is a genetic epilepsy disorder, the majority resulting from mutations in the sodium channel SCN1A gene. Sudden Unexplained Death in Epilepsy (SUDEP) occurs in up to 50% of DS patients. Risk factors for SUDEP have been identified, but mechanisms are not well understood. We hypothesize that activated astrocytes mediate post-ictal neuroinflammatory damage in the medulla. This neuroinflammatory damage is thought to disrupt medullary respiratory control centers, leading to respiratory dysfunction and sudden death. Glial fibrillary acidic protein (GFAP) is a marker of astrocyte activation and of neuroinflammation. Understanding the mechanisms of SUDEP in DS is significant as it will allow us to begin to develop therapies to prevent this fatal outcome.
Methods:
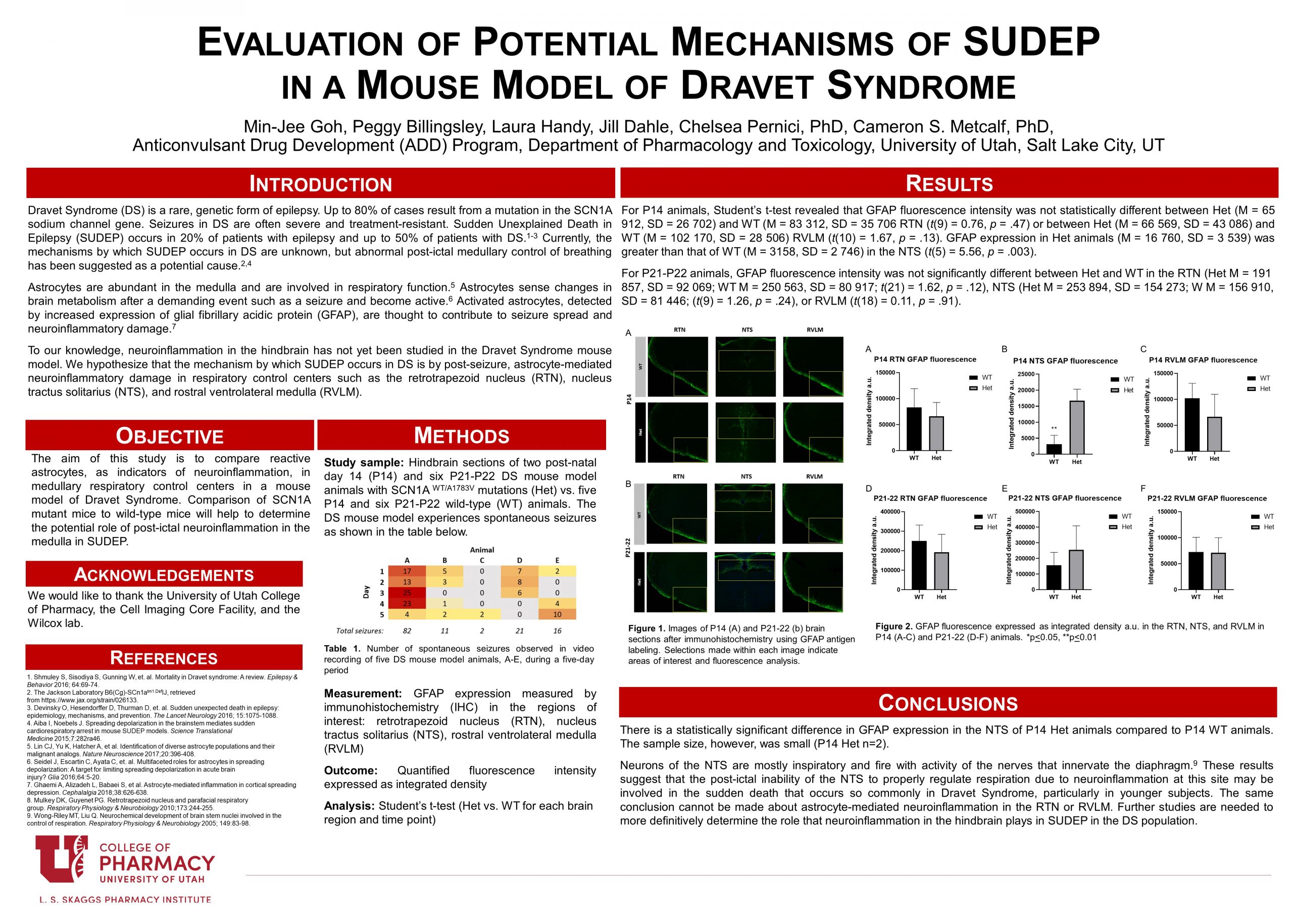
The objective of the study is to compare neuroinflammation in medullary respiratory control centers of the Dravet Syndrome mouse model (Het) to wild-type (WT) animals of two different ages, post-natal day 14 (P14) and P21-22 using GFAP expression as a marker of astrocyte-mediated inflammation. Brain tissue from WT and Het animals were sectioned and immunohistochemistry was performed to determine GFAP expression in three medullary respiratory control centers: the retrotrapezoid nucleus (RTN), nucleus tractus solitarius (NTS), and the rostral ventrolateral medulla (RVLM). Quantified GFAP fluorescence intensity was compared for Het vs. WT for each brain region and age.
Results:
There is a statistically significant difference in GFAP expression in the NTS of P14 Het animals compared to P14 WT animals. This differs from the RTN and RVLM of P14 and NTS, RTN, and RVLM of P21-22 animals, where no significant difference in GFAP expression was seen between WT and Het animals.
Conclusions:
These results suggest that the post-ictal inability of the NTS to properly regulate respiration and respond to hypoxia due to activated astrocyte-mediated neuroinflammation at this site may be involved in the sudden death that occurs so commonly in Dravet Syndrome, particularly in younger subjects. The same conclusion cannot be made about the RTN or RVLM. Further studies are needed to more definitively determine the role that neuroinflammation in the hindbrain plays in SUDEP in the DS population.

Nice work! I’m glad you were able to get some results before the lab shut down. I know the methodology turned out to be very hard to execute!
Thanks Dr. Henchey!
Nice study. It appears that your results are statistically significant yet your statements seem to suggest that you don’t believe the results. Can you provide any alternative theories as to why the results may not be accurate? Also, (a statistical question), do you need to increase your sample size to have rule out a Type 1 error in this study? Why/why not?
Dr. Malone, thank you for your questions. Although the results were statistically significant, I’m cautious about the conclusions that can be made due to the small sample size. Because a large sample size would allow us to detect smaller differences, we would need to increase the sample size to rule out a type I error.
Well done, Min-Jee. Such studies are always challenging, particularly when you’re just starting out in the lab and don’t have a lot of time to get really comfortable with the assay and analysis. So, kudos to you for getting to this point! Obviously, more will need to be done, but it was a good start. I’m curious if you have any sense as to whether the expression varies depending on the number of seizures the animal had experienced? While the sample size is going to be too small to draw a definitive conclusion, I’m just wondering if you had looked at that question in a preliminary manner.
Thank you Dr. Keefe! Because the animals that were monitored by video are not the same animals that GFAP expression was evaluated in, we can’t say how GFAP expression changes with seizure burden. That would be interesting to see and something to consider as we increase our sample size.
Hi Min-Jee, Exciting data. Could you tell me how many brain sections per animal you evaluated? Also, can you speculate what it is about the function of activated astrocytes that might be contributing to respiratory problems? Thanks! Looking forward to seeing you in the lab.
Hi Karen, I ended up having looked at one section per animal. That’s a great question. I think reactive astrocytes’ altered neurotransmission could influence their ability to support normal respiration or respond to post-ictal hypoxia. These cells may also be involved in SD silencing of respiratory control centers. Thank you and your lab for all the help with this project! I’m looking forward to your talk on these cells on Monday.
Hi Min-Jee, congratulations on your work and results. This is a very challenging project, and I hope you enjoyed this important SUDEP research.
Thank you Dr. Bulaj! I learned a lot and am so grateful to have a chance to keep learning about SUDEP.
Very nice work, Min-Jee! Your answers to comments are very thoughtful too. I wonder what is the reason for you to choose P14 and P21-21. Are these two time points special for DS in mice?
Hi Dr. Chen, thank you! You ask a good question. Dravet syndrome begins very early in life (patients often have their first seizure within the first year of life) and survival studies conducted previously by the ADD lab have shown that P14-P21 of the DS animal model is a vulnerable time so we chose to look at GFAP expression in these age groups.
Nice job!
Thank you!
Min-Jee – what an accomplishment!! I know that a project like this would be difficult to execute. I applaud all of your achievements and I am proud of what you have accomplished, starting out in the LEAP Program and then shadowing at Central City, and then all of your academic accomplishments. Well done and congratulations!
Dr. Shane-McWhorter, thank you! It’s been so great being in touch with you throughout undergrad and pharmacy school. Thanks for stopping and for your support all these years! I really appreciate it.