Relationship Between Tumor Mutational Burden and Progression-Free Survival in Non-Small Cell Lung Cancer Patients Treated with Immunotherapy
Abstract.
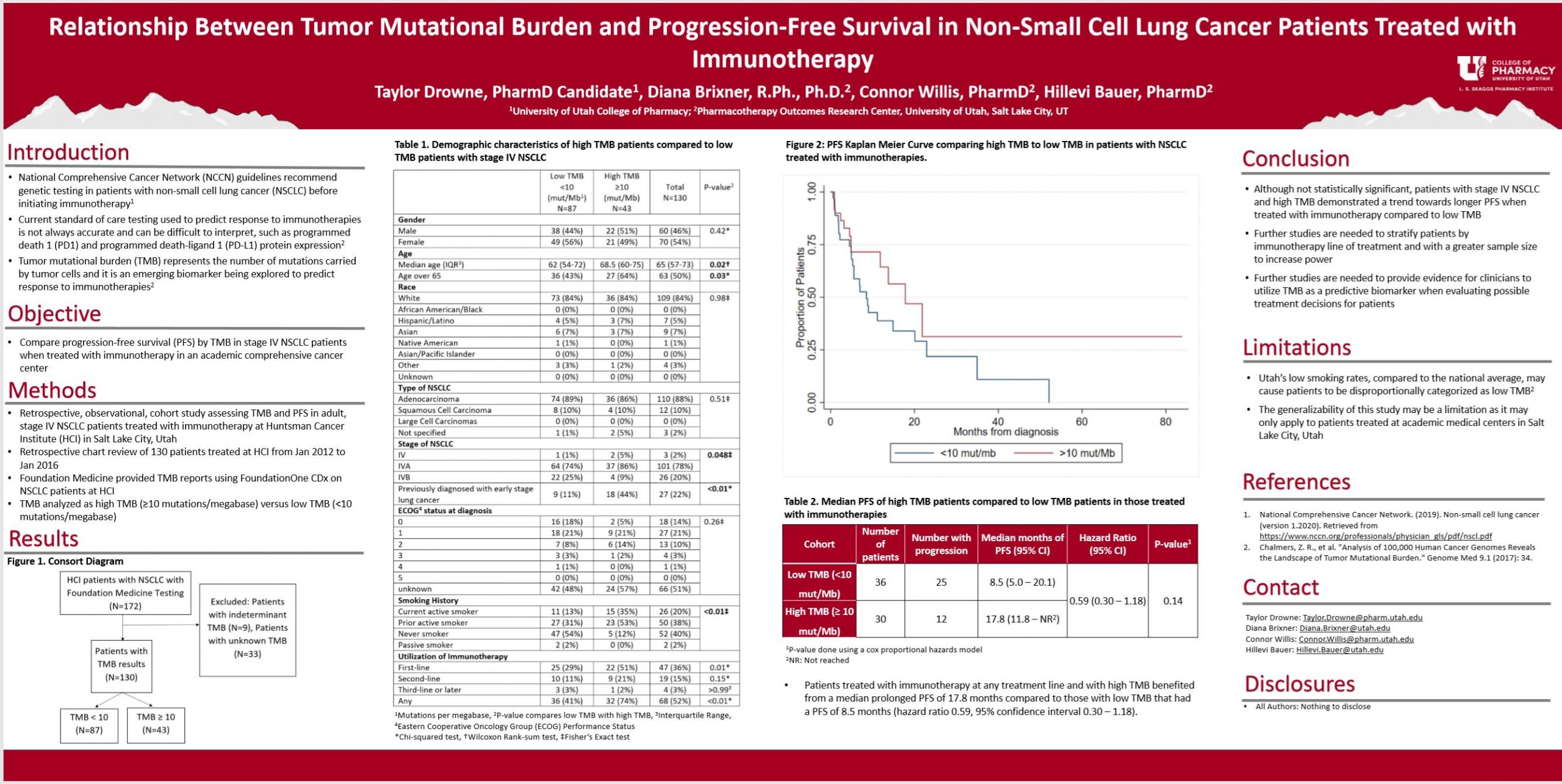
Purpose: Tumor mutational burden (TMB) is an emerging biomarker that represents the number of mutations carried by tumor cells. TMB is being explored as an independent biomarker to predict response to immunotherapies beyond the current standard of care (testing for programmed death-ligand 1 [PD-L1] expression levels). The purpose of this study is to determine if stage IV non-small cell lung cancer (NSCLC) patients with high TMB achieve prolonged progression-free survival (PFS) compared to patients with low TMB when treated with immunotherapy.
Methods: This is a single-site, retrospective, observational, cohort study assessing TMB and PFS in adult, stage IV NSCLC patients treated with immunotherapy at Huntsman Cancer Institute (HCI) in Salt Lake City, Utah. A retrospective chart review of 172 patients treated at HCI was performed. Foundation Medicine provided TMB reports using FoundationOne CDx on NSCLC patients at HCI from 1 January 2012 to 26 January 2016. PFS data was collected from the tumor registry and HCI medical chart review, and results were stratified by TMB. Kaplan-Meier curves were constructed to estimate PFS.
Results: Patients with high TMB treated with immunotherapies experienced a median PFS of 17.8 months compared to patients with low TMB that had a median PFS of 8.5 months. These results validate previous studies that explored a positive association between TMB and PFS with immunotherapies.
Conclusion: Patients with stage IV NSCLC and high TMB benefit from a prolonged PFS compared to patients with low TMB when treated with immunotherapies. Further studies are needed to provide evidence for clinicians to utilize TMB as a biomarker when evaluating possible treatment decisions for patients with stage IV NSCLC.
Published in College of Pharmacy, Virtual Poster Session Spring 2020
Taylor, great job! Any thoughts on if statistically significant Progression Free Survival (PFS) would begin to show up if we stratified TMB in increments of 10 mut/Mb? For example, a cohort that looks at 10-20 mut/Mb vs. 20-30 mut/Mb. I don’t know as much as you do about TMB or what the epidemiological data has showed as the highest TMB ever identified in a patient, but I wonder if we can find a TMB cut-off where we’ll feel really confident that the immunotherapy will be beneficial.
Hi Michael, thank you for your question. I think I understand what you are asking. I think statistical significance would begin to show up with a larger patient population. There is not currently a set standard or threshold for TMB cutoffs when it comes to mut/Mb. Most of the patients in my data set had a TMB of 20. That is why I picked 10 mut/Mb as my cutoff. With further studies, I am sure there will be a determined set cutoff value for TMB. Please let me know if I did not answer your question.
Sorry, I think my reply got messed up. Most of the patients in my data set had a TMB of 20. I am not sure the highest TMB ever identified but I have never seen >30 mut/Mb.
Most of my patients had a TMB of less than 10. Very few patients had a TMB of more than 20.
Taylor, very interesting! Sorry if my question was confusing. Either way you answered it. Thank you so much for your response!
Do you have any guesses as to why the >10MB line flat-lines in figure 2? It’s quite striking that it just stops changing after 20 months.
Hi Dr. Henchey, thank you for the question. I believe this is because we censored patients at the date of the last follow-up. Ideally, we would have added tick marks at each point a patient was censored.
Exciting results! Great job.
Hi Taylor,
This is really interesting, and counter-intuitive to me. I’d have thought that increase mutational burden would equate to increased tumor heterogeneity, and therefore reduced therapeutic response.
Hi Dr. Barrows,
The idea is that the higher mutational burden means more neoantigens that are recognized as foreign by T cells which would lead to a better antitumor response when treated with immunotherapy when compared to low TMB. Please let me know if this does not answer your question. Thank you.
Taylor, nice work! The poster is nicely laid out and clearly presents the findings. I also was surprised by the survival curves. I would have expected the opposite. Do you think that patients with greater TMB are receiving more aggressive treatment early on and, therefore, experiencing the survival benefit?
Hi Dr. Keefe,
I think that could be part of it, and that is why I would want to do a future study stratifying patients receiving different treatments as first-line for example: stratifying patients that receive immunotherapy first-line and that did not receive any chemotherapy before since my study did not do this. But the evidence and previous studies on TMB as a future biomarker are based on higher mutational burden = more neoantigens recognized as foreign by T cells which would lead to a better antitumor response when treated with immunotherapy. Please let me know if this does not answer your question. Also, the providers in this study did not use TMB to pick treatment options, so I do not think we could conclude that those with greater TMB received more aggressive treatment earlier on just because they had higher TMB. These providers were using the standard of care (the PD-L1 expression levels and targeted gene biomarkers) to determine treatment. Thank you.
Nice job on this project!